4 min read
Getting your antigen to stand out in the crowd
Here at Antibody Solutions, we believe that starting with the best possible input will yield the best possible output. A critical input for any antibody discovery program is the immunogen; i.e., what will be administered to the host animal to raise an antibody response? While it may seem complex, intact cells bearing your target antigen may be the best input. We explore that subject in this blog post through a series of frequently asked questions from our clients.
What is the advantage of using intact cells for immunization in the first place?
There are other types of immunogens that you could consider for an immunization campaign (e.g., recombinant proteins, peptides, etc.). However, the list narrows depending on the characteristics of your target and the epitope you need the antibody to recognize. Some targets (such as multi-pass membrane proteins, including G-protein coupled receptors, ion channels, and transporters) are best presented to an immune system in a natively folded state associated on a cell membrane. Other targets, including multimeric receptors or receptors requiring a chaperone are likewise best presented on a cell membrane since the antibody response will be directed to epitopes that are typically exposed in situ.
Can’t we just use our stable CHO or HEK cell line for immunization?
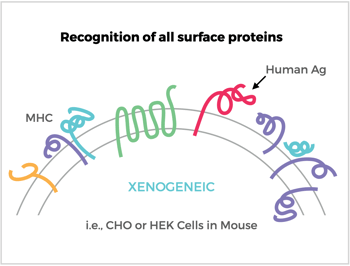
This is a question that gets asked quite frequently and, while it’s certainly possible to raise antibodies using them, there’s a whole host of issues that can arise from utilizing these types of cell lines for immunization. While it’s true that you can develop a high-expressing cell line in CHO and HEK cells, they really aren’t ideal for use as an immunogen because there is a high risk for off-target xenogeneic response in the animals. Proteins are about half the total mass of a cell membrane, and a typical cell may express over 1,000 different membrane proteins.
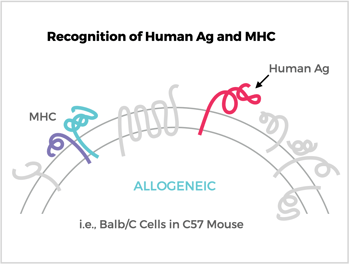
The mouse immune system will see many of the foreign proteins present on CHO and HEK and generate antibodies to them. Even immunizing with mouse cells from a different strain of the host may raise antibodies to major histocompatibility complex (MHC) proteins (i.e., allogeneic response). Using foreign antigen-bearing cells can also require a complex counter-screening strategy of multiple cell types to discern the antibodies specific for the target antigen (e.g., looking for a proverbial needle in the haystack). The bottom line is the odds are reduced for eliciting and detecting antibodies to your target protein, especially when using CHO and HEK cells.
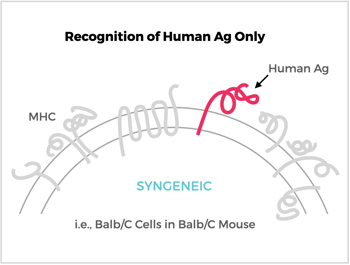
Antibody (Ab) Responses to Surface Antigens (Ag) on Recombinant Ag-Expressing Cells
If intact cells are the best option for immunization, then what are the best cells for presenting my target?
This is where the generation and use of syngeneic cells provides the best input for presenting your target. Syngeneic cells are cells that are derived from the same genetic background as the particular species and strain of rodent that will be used for immunization. The immune system of a syngeneic host is essentially blind to self-like membrane proteins. By contrast, your target antigen will “stand out” on cells and give it the best chance of eliciting an antibody response.
We have identified a panel of syngeneic cells that can give quite high levels of membrane protein expression, similar to or better than CHO or HEK cells. But the biggest advantage is, because they are derived from the same species and strain, you don’t have the risk of making off-target antibodies. An additional benefit is that the transfected syngeneic cells can be used for antibody screening assays.
So, how do I get syngeneic cells expressing my protein?
Engineered syngeneic cells are obtained by transfection with the target protein followed by antibiotic selection. Our approach utilizes optimized vectors for expression and our flow cytometry expertise to pick out the highest expressing clones. We have identified syngeneic cell lines that can be used specifically for immunization of rats and multiple strains of mice. In addition, we’ve expressed hundreds of proteins in mouse and rat cells, including difficult ones like multipass and multimeric membrane proteins.
Surface Antigen Cell Expression Workflow
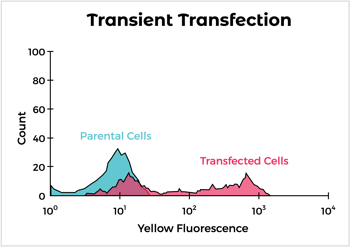
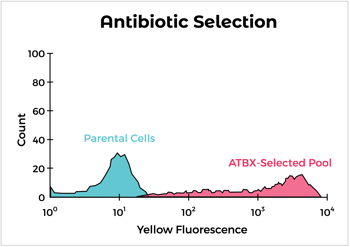
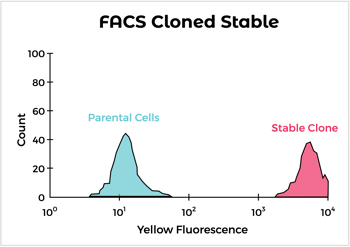
But what if I don't have time to wait for cell line development?
While it can take a few months to obtain a stable clone, if time is of the essence, we can generate antibiotic-selected pools of cells expressing high levels of target antigen in as few as four weeks. These can be used for initial immunizations while obtaining a stable clone for later boosts and screening assays. In addition, we can use the vector validated by syngeneic cell expression for DNA immunizations.
Other research you might be interested in:
 We also touch on the pros and cons of using different types of immunogens (including transfected cells) in our poster, “Generation of Antibodies to Difficult Membrane Protein Targets,” so that you can see how the cells compare to other methods.
We also touch on the pros and cons of using different types of immunogens (including transfected cells) in our poster, “Generation of Antibodies to Difficult Membrane Protein Targets,” so that you can see how the cells compare to other methods.
Ready to Speak with an Expert?
Let’s have a conversation to determine which cell line or lines would work best for your projects and help both you and your antigens stand out in the crowd!

Michael works closely with his clients to help them navigate their research studies from start to finish. His responsibilities include helping clients select the most promising strategy to use to yield their desired results and then managing the work in an efficient and cost-effective manner.



